Japan

Ginkgo Bioworks and Sumitomo Chemical Announce Expanded Partnership To Develop Functional Chemicals with Synthetic Biology
Sumitomo Chemical Co., Ltd (TSE:4005), one of Japan's leading chemical companies, and Ginkgo Bioworks (NYSE: DNA), which is building the leading platform for cell programming and biosecurity, today announced a new program to develop functional chemicals with synthetic biology and expand upon the companies' existing biomanufacturing partnership.

Astellas and Eko Health Announce Agreement to Incorporate Next Generation Eko CORE 500™ Digital Stethoscope into Z1608, an Innovative Solution Under Development for Heart Failure Patients Built on Welldoc’s Proven Digital Therapeutic Platform
Astellas Pharma Inc. (TSE: 4503, President and CEO: Naoki Okamura, "Astellas") and Eko Health Inc. (Co-Founder and CEO: Connor Landgraf, "Eko") announced today that they have entered into a License & Supply Agreement for the global supply and license of Eko's latest digital stethoscope, the CORE 500™, and a custom suite of Eko's AI-powered cardiovascular disease detection software.

European Medicines Agency Accepts Astellas’ Marketing Authorization Application for Zolbetuximab
Astellas Pharma Inc. (TSE: 4503, President and CEO: Naoki Okamura, "Astellas") today announced the European Medicines Agency (EMA) has accepted for regulatory review the company's marketing authorization application (MAA) for zolbetuximab.

JEOL: Release of Scanning Electron Microscopes JSM-IT710HR/JSM-IT210
JEOL Ltd. (TOKYO:6951) (President & CEO Izumi Oi) announces the release of scanning electron microscopes JSM-IT710HR/JSM-IT210 on July 23, 2023.

Eisai to Present the Latest Alzheimer’s Disease Pipeline and Research, including Lecanemab and Anti-MTBR TAU Antibody E2814, at the Alzheimer’s Association International Conference 2023
Eisai Co. Ltd (Headquarters: Tokyo, CEO: Haruo Naito, "Eisai") announced today that the company will present the latest findings on its Alzheimer's disease (AD) pipeline and research, including Eisai's anti-amyloid beta (Aβ) protofibril* antibody for the treatment of Alzheimer's disease (AD), lecanemab (generic name, U.S. brand name: LEQEMBI®), and the company's investigational anti-MTBR** tau antibody, E2814, at the Alzheimer's Association International Conference (AAIC).

Sosei Heptares Webinar Presentation for Q2 FY2023 Financial Results
Sosei Group Corporation (“the Company”; TSE: 4565) will announce its earnings results and present operational highlights for the 6 months ended 30 June 2023 on Friday, 4 August 2023.

Astellas and 4D Molecular Therapeutics (4DMT) Enter into License Agreement to Use 4DMT’s Proprietary Intravitreal R100 Vector for Rare Ophthalmic Targets
Astellas Pharma Inc. (TSE: 4503) (President and CEO: Naoki Okamura, "Astellas") and 4D Molecular Therapeutics, Inc. (NASDAQ: FDMT) (CEO: David Kirn, MD, "4DMT") today announced a license agreement under which Astellas gains rights to utilize the intravitreal retinotropic R100* vector invented by 4DMT for one genetic target implicated in rare monogenic ophthalmic disease(s), with options to add up to two additional targets implicated in rare monogenic ophthalmic diseases after paying additional option exercise fees.

Sosei Heptares Doses First Subject in Phase I Trial with HTL0048149, a First-in-Class GPR52 Agonist for Schizophrenia
Sosei Group Corporation (“the Company”; TSE: 4565), the world leader in G protein-coupled receptor (GPCR) focused structure-based drug design (SBDD) and development, announces it has dosed the first subject in a Phase I trial evaluating HTL0048149 (HTL’149), a first-in-class GPR52 agonist, which represents a novel mechanism of action for the treatment of schizophrenia and related neurological diseases.
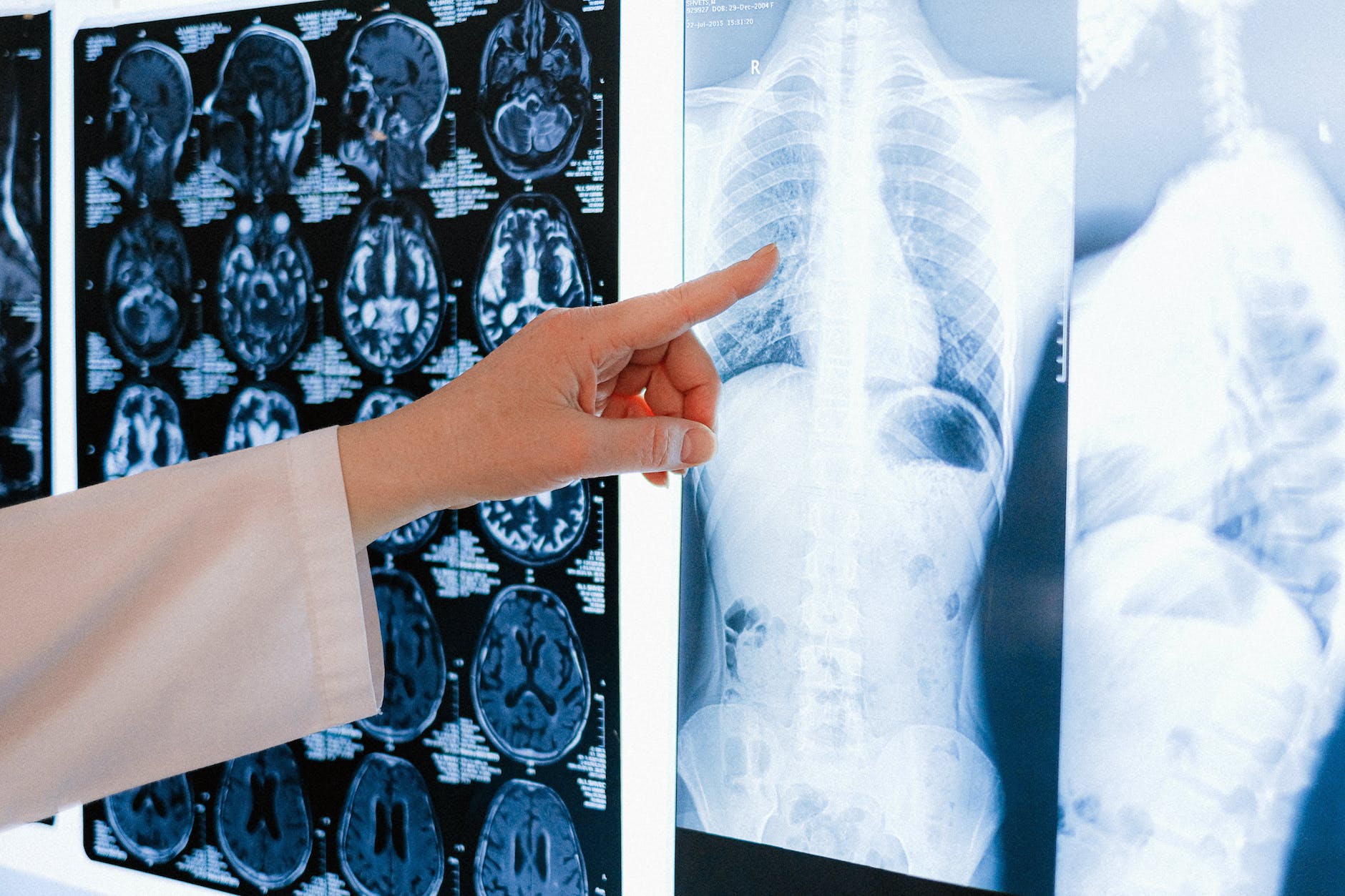
Datopotamab Deruxtecan Met Dual Primary Endpoint of Progression-Free Survival in Patients with Advanced Non Small Cell Lung Cancer in TROPION-Lung01 Phase 3 Trial
Topline results from the TROPION-Lung01 phase 3 trial showed datopotamab deruxtecan (Dato-DXd) demonstrated a statistically significant improvement for the dual primary endpoint of progression-free survival (PFS) compared to docetaxel, the current standard of care chemotherapy, in patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) treated with at least one prior therapy.

Astellas Announces U.S. FDA Grants Priority Review for Zolbetuximab Biologics License Application
Astellas Pharma Inc. (TSE: 4503, President and CEO: Naoki Okamura, "Astellas") today announced that the U.S. Food and Drug Administration (FDA) has accepted and granted Priority Review for the company's Biologics License Application (BLA) for zolbetuximab, a first-in-class investigational Claudin 18.2 (CLDN18.2)-targeted monoclonal antibody, for first-line treatment of patients with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors are CLDN18.2-positive.







