South Korea

Novotech Presents Technical Session on Clinical Trial Success Factors at BIOPLUS-INTERPHEX KOREA 2023
Novotech, the leading Asia Pacific centred biotech CRO with global execution capabilities will present a technical session at BIOPLUS-INTERPHEX KOREA 2023 for biotechs planning regional or global trials.
Lunit’s AI-Powered Lung Cancer Screening Solution Significantly Affects Radiologists’ Diagnostic Determination – Published in Radiology
Lunit (KRX:328130.KQ), a global provider of AI-powered cancer diagnostics solutions, today announced the publication of a study exploring the impact of medical AI solutions' accuracy on radiologists' diagnostic determination.

Korea University Medicine Study Highlights a New Biomarker for Major Depressive Disorder
Researchers led by Professor Byung-Joo Ham and Associate Professor Kyu-Man Han from Korea University Medicine have successfully identified a neuroimaging-based biomarker for MDD in a recent study published on 8 May 2023 in Psychological Medicine.
Zymedi announced their collaboration with the National Heart, Lung, and Blood Institute through a CTA CRADA to develop ZMA001 mAb, a potential treatment for Pulmonary Arterial Hypertension (PAH), a rare, female predominant disease
Zymedi, a Korean biotech venture-backed company, announced the signing of a clinical Cooperative Research and Development Agreement (CRADA) with the National Heart, Lung, and Blood Institute (NHLBI), part of the National Institutes of Health (NIH), to develop ZMA001 for the treatment of pulmonary arterial hypertension (PAH).
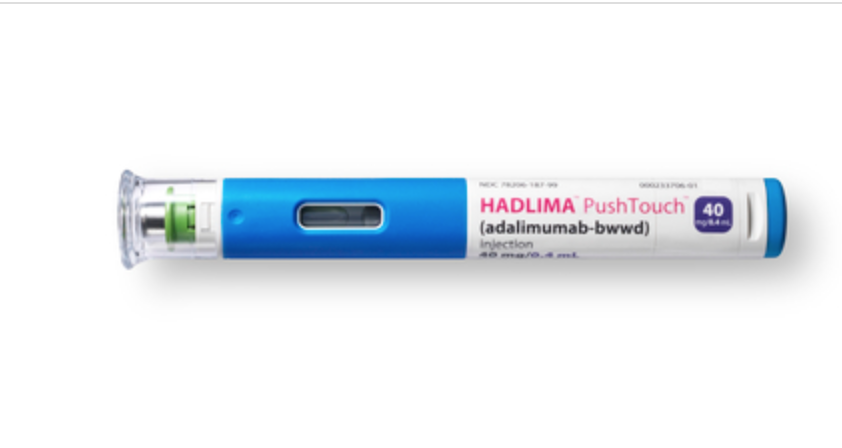
REPEAT/Organon & Samsung Bioepis Announce US Launch of HUMIRA Biosimilar HADLIMA™ (adalimumab-bwwd) in Multiple Presentations Consistent with Originator
Organon & Co. (NYSE: OGN) & Samsung Bioepis Co., Ltd. announced that HADLIMA™ (adalimumab-bwwd), a biosimilar referencing Humira (adalimumab), is now available to patients in the United States. Consistent with Humira, HADLIMA is available in both citrate-free high concentration (100 mg/mL) and citrate-containing low concentration (50 mg/mL) to provide patients with seamless continuity of care.

TiumBio and Hansoh Pharma Announce Clinical Trial Approval of ‘HS-10518/TU2670’ from NMPA in China
TiumBio Co., Ltd. (KOSDAQ: 321550), a clinical-stage biopharmaceutical company focusing on discovering and development innovative therapeutics for patients with rare and incurable diseases, and Hansoh Pharmaceutical Group Co., Ltd. (SEHX: 3692), a leading biopharmaceutical company, announced that the China National Medical Products Administration (NMPA) approved Hansoh's Investigational New Drug (IND) application for clinical study of HS-10518 (also known as TU2670), the management of moderate to severe pain associated with endometriosis.

SK bioscience- Government Pharmaceutical Organization Sign Memorandum of Understanding to Strengthen Vaccine Infrastructure in Thailand
SK bioscience announced that the company has signed a Memorandum of Understanding (MOU) with the Government Pharmaceutical Organization (GPO), a state pharmaceutical enterprise in Thailand, to strengthen Thailand's vaccine infrastructure.

MEDICAL IP Received US FDA 510(k) Clearance for DeepCatch, AI Software for Whole-Body Composition CT Quantification
MEDICAL IP (CEO Joon S. Park), an AI-based digital twin company, announced that CT-based automatic body composition analysis AI software DeepCatch received US FDA 510(k) Clearance.

Kalos Medical Receives Korean MFDS Innovative Medical Device Designation for DENEX Hypertension Treatment System
Kalos Medical Inc. announced that South Korea’s Ministry of Food and Drug Safety designated the company’s DENEX hypertension treatment system as an Innovative Medical Device.
SK bioscience and the Peter Doherty Institute for Infection and Immunity Join Forces Against Influenza
SK bioscience, a global innovative vaccine and biotech company committed to promoting human health from prevention to cure, announced that the company has signed a research collaboration agreement on influenza research with the Doherty Institute.







